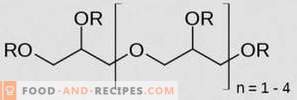
Sodium salts of orthophosphoric acid, also known as sodium orthophosphates, have long been successfully used in the food industry. On many foods today, you can read that they contain E339 - a stabilizer, emulsifier and antioxidant in one person, which is a colorless or white crystalline powder. Sodium orthophosphates dissolve well in liquids, creating an alkaline environment, have high resistance to high temperatures and at the same time can enhance the properties of natural antioxidants.
What is the benefit of E339 and where is this supplement used?
Initially, sodium orthophosphates were widely used in the chemical industry, being an integral part of all kinds of detergents and cleaning products. However, practice has shown that after the end of the chemical reaction, an insoluble precipitate forms in the water, and thus the additive E339 becomes a source of serious environmental problems. It is for this reason that many countries in the world today have refused to add sodium orthophosphate to detergents, however this substance is still used very actively in the food industry. First of all, E339 is added to pastries, sweets and candies to minimize oxidative processes. For the same reason, sodium orthophosphates today are part of many dairy products, which increases their shelf life at least twice. It is also worth noting that E339 contain various semi-finished products and, in particular, dry breakfasts, soups in bags, granulated teas, when steamed, the task is to obtain a homogeneous mass of substances that often contact. In addition, to preserve the freshness and natural taste, sodium orthophosphates are often added to meat and fish, as well as various types of canned food. Meet this stabilizer can be in some carbonated drinks, which are replete with chemical dyes and all sorts of food additives.
Why are sodium orthophosphates harmful to humans?
Using the example of environmental pollution with insoluble precipitations, it is not difficult to assume that something similar happens in the human body. This is true because the by-products obtained by contact of various substances with E339 turn into slags, which are not so easy to get rid of. In addition, sodium orthophosphates themselves are very harmful to human health, as they contribute to the leaching of potassium from the body. As a result, the teeth and nail plates are destroyed, and the bones become fragile and brittle. Foods that contain sodium orthophosphate can cause disturbances in mineral metabolism in the body or cause serious problems with digestion, including prolonged diarrhea. It has also been proven that sodium orthophosphates are a fairly potent carcinogen, and the consumption of food containing such an additive can lead to the development of cancer. However, the maximum allowable dose of the content of E339 in food has not yet been determined.