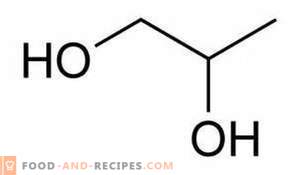
Very often, when buying cosmetics or food, a substance such as propylene glycol is indicated on their label. It is usually encrypted as an E1520 preservative, considered to be relatively safe. There are two types of propylene glycol, the first of which is really toxic and is intended for use in the chemical industry in the production of polyester resins. The second type of propylene glycol is used in the food and cosmetic industry. This “eshka” has a sweetish taste and a subtle luscious smell, which is easily suppressed by other flavors. Therefore, to determine the presence of E1520 in a particular product without laboratory research is almost impossible.
Field of application of propylene glycol
Initially, this colorless liquid, which has long been learned to be produced by synthesis, has found its application in cosmetics and perfumery. Propylene glycol served not only as a preservative, but also as an excellent moisturizer. By binding water molecules, he prevented them from exiting through the skin pores naturally. This principle is based not only on the action of moisturizers, but also on modern deodorants, which, as advertising says, can clog the sweat glands for 24 hours.
Much later, propylene glycol was used in the food industry to preserve the taste of products. In particular, this additive can be found in some types of vegetable oils, in animal feed, artificial vegetable cream and all kinds of canned foods. Recently, it has been added to tobacco, as well as to e-cigarette liquid, in order to improve the taste of smoke. Elixirs for rinsing the mouth, liquid for automotive glass that prevents them from freezing, mixtures of anti-icing for aviation, heating systems and air conditioning systems - this is not a complete list of areas in which propylene glycol has found quite wide application.
Useful properties of propylene glycol
In addition to preserving taste and ability to retain moisture, propylene glycol is able to influence the temperature of the liquid. This property turned out to be very popular when it became necessary to extend the shelf life of perishable products. By creating a cooling effect, propylene glycol acts on the principle of a portable refrigerator. The only difference is that it does not require power. But the principle of the E1520 is really similar to the work of the freezer. True, the temperature in this case will be different. However, practice shows that even insignificant cooling of products that deteriorate in a heat in a few hours can preserve their attractive appearance and excellent taste for a longer time.
Nowadays, propylene glycol is obtained from petroleum products, however this chemical substance does not accumulate in the body and is easily excreted naturally. Moreover, it is able to enhance the effect of medicines, therefore polypropylene glycol is often included in the composition of modern drugs.
What is dangerous propylene glycol?
When it comes to cosmetics, most people can use them without special concerns, because the content in them of such a preservative as E1520 is minimal. However, occasionally, there are still cases when an innocent lotion or face cream causes severe allergies. This is due, as a rule, to individual intolerance to propylene glycol, which occurs infrequently.
The situation is much more complicated when you buy sprays or hair sprays, which include this substance. The thing is that propylene glycol fumes have a very negative effect on the respiratory organs and can provoke asthma attacks. This is especially dangerous for people who suffer from asthma or tuberculosis. Shampoos with E1520 are not always safe to use, as this substance may cause conjunctivitis if it comes into contact with the mucous membrane of the eyes. As for the blockage of sweat glands, this procedure is not so harmless as it seems at first glance, and can lead to serious inflammatory processes. In foodstuffs, propylene glycol is much less common than in cosmetics. However, you should not use these "delicacies" to people who suffer from diseases of the heart and gastrointestinal tract.
Medical studies have shown that the limiting dose of a substance such as propylene glycol in the blood is 1 gram per 1 l. However, to do this, the colorless liquid must either be drunk in large quantities or administered intravenously. If this happens, death from cardiac arrest is inevitable. With a lower concentration of the substance in the blood, the development of bradycardia, hypotension, hay fever, hymolysis and lactic acidosis are possible.